 After installing the doser on the ultrahigh vacuum chamber, an electrical short was detected. There have been a few challenges during Year 2. For these calculations, 2 surface oscillators were assumed and a low frequency of 200 cm -1. MURT predictions of the dissociative sticking coefficient as a function of reaction threshold energy (E 0). The image on the left shows the bare doser, while the image on the right shows it ready to be attached to the UHV chamber.įigure 3. Photographs of the variable temperature effusive molecular beam built in the Abbott-Lyon lab. Schematic diagrams of the variable temperature effusive molecular beam, viewed from the top (left) and from the rear (right), were created in LibreCAD.įigure 2. Additionally, the MURT code was updated for CH 4/V(100) and sticking coefficient predictions were made assuming a range of conditions based on other CH 4/metal systems that have been studied.įigure 1. In order to perform the dissociative sticking experiments, a heated effusive molecular beam doser was designed and built during Year 1 of this award (Figures 1 and 2). These non-equilibrium measurements will be analyzed using a microcanonical unimolecular rate theory (MURT) to extract the reaction threshold energy (E 0) and other characteristics of the transition state such as the contribution from the surface. 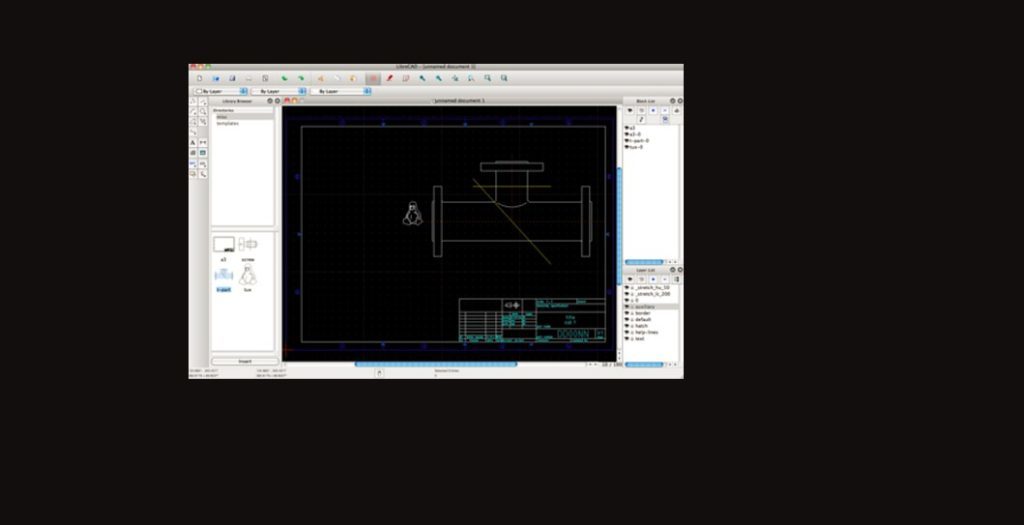
In particular, methane dissociative sticking coefficients will be measured as a function of independently varied gas and surface temperatures. Using experimental and theoretical methods to investigate the activity of bare and oxygen pre-covered vanadium surfaces, the Abbott-Lyon Lab (ALL) will investigate the direct conversion of methane to methanol. We hypothesize that a methoxy intermediate, similar to the one observed for CH 3OH to CH 2O conversion, may be important in the partial oxidation of methane to methanol. Vanadium oxide catalysts are known to be effective for the partial oxidation of methanol to formaldehyde. While a direct conversion method would be preferred, it is difficult to perform partial oxidative dehydrogenation rather than complete oxidation to CO 2 because of the reaction thermodynamics.  
Current methods for methane conversion to liquid hydrocarbons involve an indirect two-step process: 1) syngas (CO + H 2) is produced via oxidation of methane and 2) a Fischer-Tropsch reaction is used to create a hydrocarbon. Converting methane into partially oxidized compounds such as methanol and formaldehyde, which are important feedstock compounds in the chemical industry, is highly desirable. Methane is an abundant resource within the United States. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
